This is an accordion element with a series of buttons that open and close related content panels.
HIV Reservoirs: How Low Must We Go?

An essential question for HIV cure research is: how small must HIV reservoirs be to achieve sustained remission without antiretroviral drugs? While antiretroviral therapy (ART) effectively controls HIV replication, it’s not a cure. Significant efforts are focused on developing treatments to reduce viral reservoirs and enhance antiviral immunity—but exactly how small must these viral reservoirs become to allow patients to safely discontinue ART?
To address this question, we developed an innovative model in rhesus macaques that allows us to precisely control the size of latent SIV reservoirs. We generate latently infected cells in the laboratory, transfer defined numbers into ART-treated animals, and then monitor what happens when treatment stops. By using a genetically barcoded virus, we can determine not only when viral rebound occurs, but also how many individual cells are reactivated to cause it. This level of precision is difficult to achieve in human clinical trials, where reservoir sizes vary widely and are challenging to measure. Our goal is to define the reservoir thresholds that could enable people living with HIV to safely stop daily medication.
Isolating Monoclonal Antibodies

We developed a SMART-RACE-based method for cloning antibodies from individual B cells. Unlike traditional PCR approaches, which struggle with diversity in immunoglobulin leader sequences, our technique eliminates the need for complex multiplex primer sets, thereby improving recovery rates from single cells.
Using this platform, we have isolated monoclonal antibodies against primate immune molecules, including macaque major histocompatibility complex (MHC) class I and killer immunoglobulin-like receptors (KIRs). Our anti-MHC antibodies are available through the NIH Nonhuman Primate Reagent Resource.
Our method works with virtually any species—from mice and nonhuman primates to ferrets, hamsters, dogs, and pigs. We offer antibody isolation services through the WNPRC Immunology Services Unit, helping researchers develop novel reagents for diverse applications.
Harnessing Wisconsin's Dairy Heritage for Drug Discovery
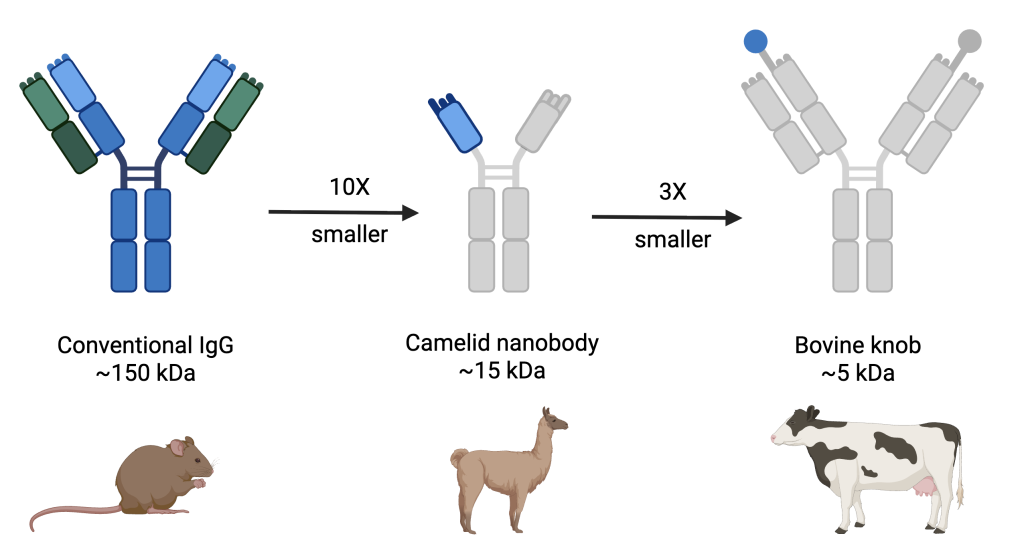 Cows are among the few species that produce antibodies with a unique architecture: an ultralong loop that forms a distinctive mushroom-like structure—a stalk topped by a compact “knob.” When isolated from the parent antibody, these knob domains are the smallest known functional antibody fragments (~5 kDa)—smaller than camelid nanobodies, shark VNAR domains, and far smaller than conventional IgG antibodies (~150 kDa). Despite their size, knob domains can achieve high binding affinities and excellent stability.
Cows are among the few species that produce antibodies with a unique architecture: an ultralong loop that forms a distinctive mushroom-like structure—a stalk topped by a compact “knob.” When isolated from the parent antibody, these knob domains are the smallest known functional antibody fragments (~5 kDa)—smaller than camelid nanobodies, shark VNAR domains, and far smaller than conventional IgG antibodies (~150 kDa). Despite their size, knob domains can achieve high binding affinities and excellent stability.
The ultra-small size of knob domains offers key advantages. They can access recessed or hidden binding sites that are out of reach to conventional antibodies, and they may penetrate dense tissues—such as the dense tissue surrounding solid tumors, which can restrict larger antibody formats. These properties make them attractive candidates for therapeutics where traditional antibodies struggle.
We are establishing a bovine antibody discovery platform at UW-Madison, adapting our proven B cell cloning methods to isolate knob domains from immunized cattle. Our current projects focus on targeting cancer antigens overexpressed on solid tumors and infectious disease targets. By harnessing Wisconsin’s dairy heritage, we aim to develop these knob domains as therapies against challenging diseases in both animals and humans.
Mapping Minor Histocompatibility Antigens
 Minor histocompatibility antigens (mHAgs) are MHC-bound peptides on cell surfaces that can stimulate immune responses after organ transplants, arising from genetic differences between donors and recipients. Identifying mHAgs is challenging due to the size of the genome and the number of potential differences between individuals.
Minor histocompatibility antigens (mHAgs) are MHC-bound peptides on cell surfaces that can stimulate immune responses after organ transplants, arising from genetic differences between donors and recipients. Identifying mHAgs is challenging due to the size of the genome and the number of potential differences between individuals.
We developed an innovative approach to map mHAgs in Mauritian cynomolgus macaques using a combination of cellular immunology, genomics, and bioinformatics. This work led to the identification of the first nonhuman primate mHAg, which now serves as a model antigen for testing mHAg-targeted therapies for blood-borne cancers and advancing HIV cure strategies in macaques.
